Smarter oncology second opinion — AI-powered, expert-reviewed, using your existing NGS report.
Read More

Molecular tumor boards (MTBs) sit at the center of precision oncology, where complex genomic data is translated into treatment decisions. Yet despite widespread adoption of sequencing, therapy selection remains inconsistent, particularly across heterogeneous cancer types.
Digital Drug Assignment (DDA), commercially known as Genomate, was designed for exactly this setting: to support MTBs by ranking therapies based on each patient's full molecular context, using computational reasoning.
New data presented at AACR 2026 demonstrate that Genomate can consistently stratify treatment efficacy based on data from a real-world molecular tumor board, across a diverse pan-cancer population. The study, titled “Clinical efficacy of computational reasoning for personalized treatment planning in a pan–cancer cohort discussed by a French multidisciplinary tumour board: a real-world experience-based analysis,” analyzed patients discussed at the Institut Curie MTB between 2018 and 2022. The collaboration underlying this work was supported by the Eurostars program (Project no. 3091, POP-GNC), enabling cross-institutional evaluation of computational reasoning in clinical practice.
Presenter: Istvan Petak, MD, PhD, Founder & Chief Scientific Officer, Genomate Health
Section: Late-Breaking Research: Clinical Research 2
Poster Number: LB129
Date & Time: April 20, 2026, 9:00 AM - 12:00 PM
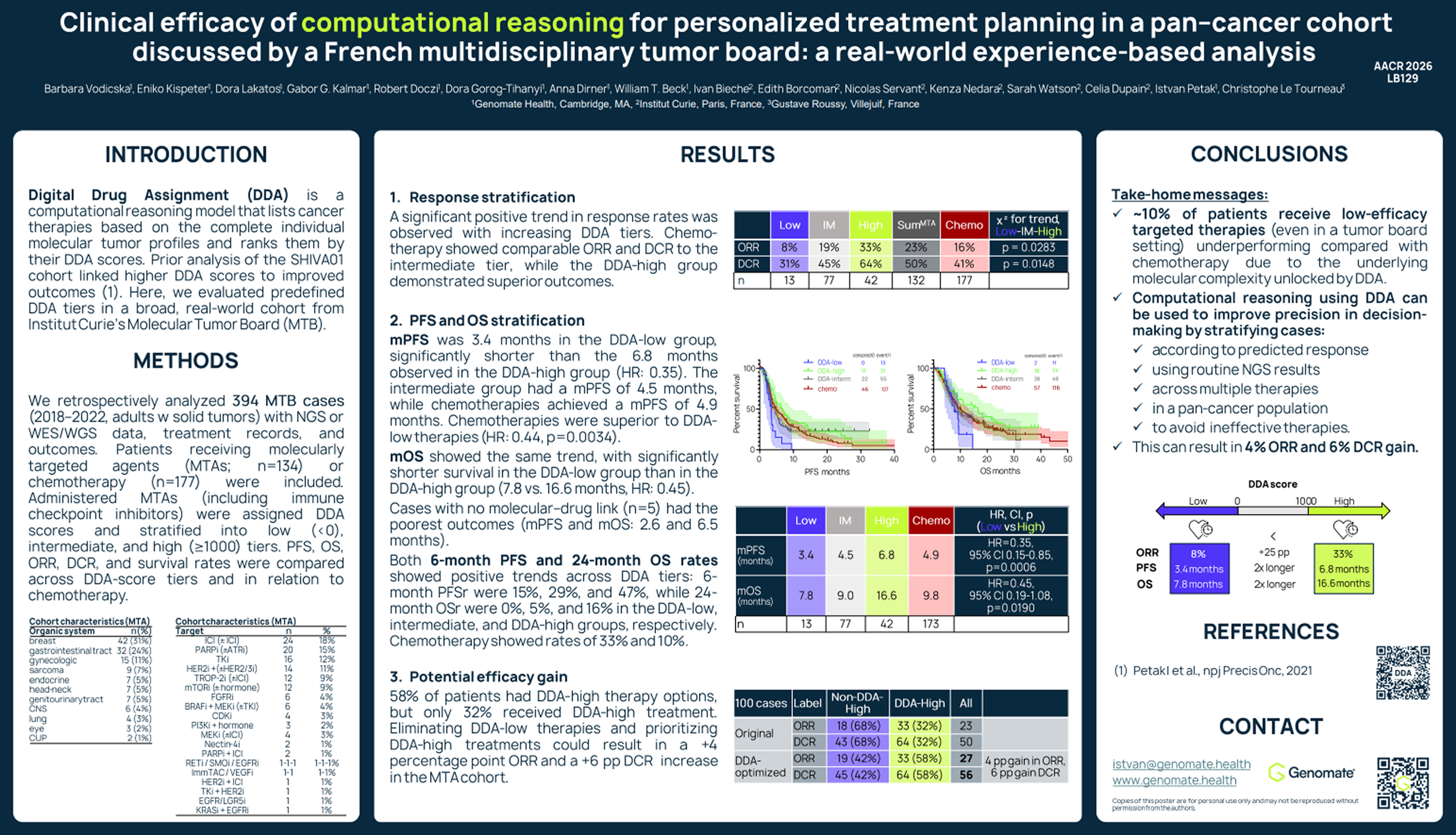
This retrospective analysis included 394 patients with solid tumors who were evaluated in routine MTB practice. Patients received either molecularly targeted agents (including immunotherapies) or chemotherapy, based on clinician decisions.
Administered therapies were retrospectively scored using DDA and grouped into predefined tiers: low, intermediate, and high. The results show a clear and consistent relationship between DDA scores and clinical outcomes:
Importantly, low-DDA therapies underperformed chemotherapy, reinforcing the model’s ability to distinguish when targeted therapy is truly beneficial and when it is not. Patients with no detectable molecular–drug link experienced the poorest outcomes, underscoring the importance of meaningful biological matching.
MTBs operate in a highly complex environment, where variability in interpretation can directly impact patient outcomes. This study shows that computational reasoning can introduce structure and consistency into treatment selection by:
Because these results are derived from real-world clinical decisions across multiple cancer types, they demonstrate that Genomate captures robust, biologically grounded treatment–outcome relationships.
This analysis highlights a critical gap in real-world MTB decision-making. While 58% of patients had high-DDA therapy options available, only 32% received them, indicating that optimal treatments are not consistently selected in practice. At the same time, approximately 10% of patients received low-efficacy targeted therapies, which underperformed compared to chemotherapy, even within an expert tumor board setting.
These findings reflect the inherent difficulty of interpreting complex molecular profiles across diverse cancer types. By retrospectively optimizing treatment selection (eliminating low-DDA therapies and prioritizing high-DDA options), the model suggests that outcomes could be improved using the same available therapies, resulting in a +4 percentage point increase in objective response rate (ORR) and a +6 percentage point increase in disease control rate (DCR) in the targeted therapy population.
Together, these results show that computational reasoning can enhance MTB decision-making by systematically identifying both effective and ineffective therapies, enabling more consistent, high-quality treatment selection in real-world precision oncology.
This research will be presented at the AACR Annual Meeting 2026 as a late-breaking abstract poster. It is part of a broader set of studies from the Genomate Health research team exploring how computational reasoning can support precision oncology. Two additional abstracts from our team will also be presented at the conference:
Regular abstract: Molecularly Informed Prediction of Treatment Efficacy in GDSC Cell Line Data Using Computational Reasoning ↗
Regular abstract: Molecularly-informed prediction of treatment efficacy in the GENIE BPC NSCLC cohort using computational reasoning ↗